|
Lattice energy=Heat of formation-Heat of atomization- Dissociation energy. Sum of Electron affinities+ Lattice energy)Īrranging the above equation to find lattice energy, we get The heat of formation=Heat of atomization+Dissociation energy of Ionization energies+ The Born-Haber cycle representation in a single equation is given below: Instead, lattice energies are determined through experimentally specific enthalpy shifts from a cycle called Born–Haber cycle. But, the estimated number of ions will never be assembled under the circumstances for determining heat transfer rate. We have some theories that the lattice energy can be calculated by adding positive and negative ions for forming an ionic solid. 
This article will show you the lattice energy equation, lattice energy formula, how to calculate it step by step, and give you examples of how the equation & formula can be utilized. 
Here, the energy that is provided to 1 mole of NaCl to isolate it into gaseous Na + and Cl – ions is 786.0 kJ. In the given ionic molecule below, the energy needed for the following reaction is the lattice energy. A sodium chloride molecule’s crystal lattice is depicted below the structure in the crystalline lattice. As a result, ions with different opposite charges tend to have energy because of electrostatic solid force. Such characteristics help ions maintain their consistent form in the crystalline lattice. Also, at higher temperatures, they tend to melt.įor instance, NaCl melts at 801 degrees Celsius. Such ionic compounds are typically crystalline, stiff, and breakable substances with smooth lattice planes. We can also notice a transfer of negative charge particles called an electron from metal to non-metal. Keeping in mind that the association between a metal and a non-metal usually results in developing an ionic compound. As gaseous ions combine to form atoms, the energy required for the conversion value is expressed as a kJ/mol unit. Since the reaction is exothermic in certain conditions, the value of the lattice energy is always negative. While we expect a systematic system to be less unchanging since its entropy is limited, the situation is not the same. Second, this energy is used in the reverse phase in which gaseous ions bond to form the atoms of an ionic solid. This description gives meaning to the lattice energy, which is always positive because the reaction is endothermic. One is the energy necessary for breaking an ionic solid as its atoms are converted to ions (gaseous). Lattice Energy definition can be explained in two distinct ways by a sort of potential energy. Generally, it is expressed as kilojoules per mole (kJ/mol). Even so, it can be calculated with the help of the Born-Haber cycle. In an Ionic solid, lattice energy cannot be directly measured. It gives insights into various characteristics, including its volatility, solubility, and durability of ionic solids. Also, it can be described as a method of measuring cohesive forces that bind ions. Lattice energy is a calculation of ionic bond strength in an ionic compound. 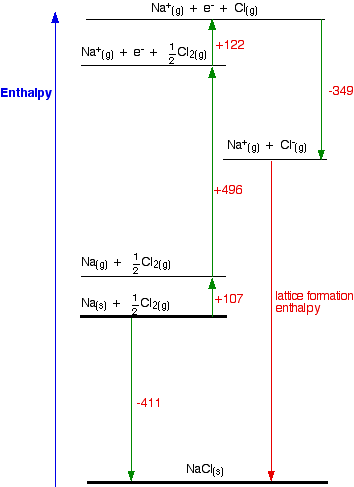
This article will elaborate only on lattice Energy, Lattice Energy definition, Lattice energy formulas, Lattice Energy equation, and some practical examples to understand Lattice Energy’s concepts. On the other hand, Lattice Enthalpy is considered the change in enthalpy whenever the bond is formed or broken. Lattice energy is the amount of energy that is required to break a solid bond into gaseous elements. However, the difference between them is negligible. These terms are differentiated based on their mode of calculation. The fact is that Lattice energy and Lattice Enthalpy has a different meaning. Both are used to calculate bond strength in an ionic or Covalent bond that ultimately elaborates the attractive forces in ions, so these terms are problematic. Another way round, some Students Consider it, they have a different meaning. Some students might consider it they have the same meaning. In Chemistry, Lattice energy and Lattice enthalpy are the two most commonly used terminologies.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
